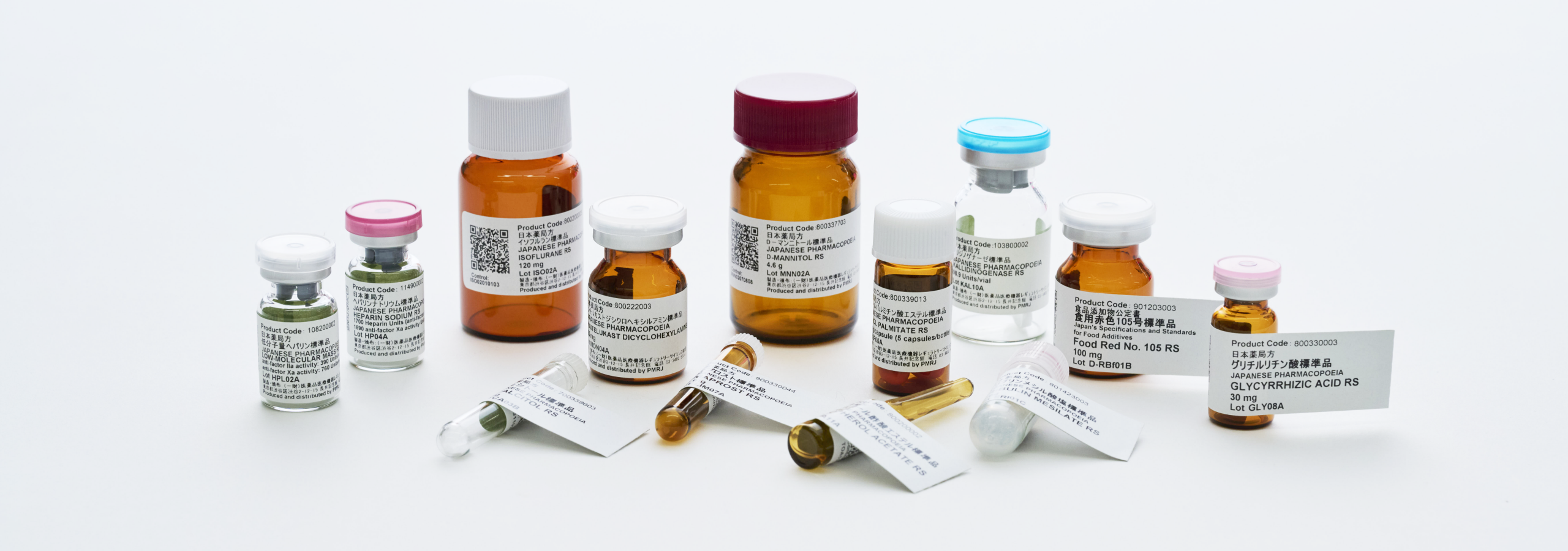
Contributing to the Assurance and Enhancement of the Quality of Pharmaceuticals
Contributing to the Assurance and Enhancement of the Quality of Pharmaceuticals
PMRJ produces and distributes compendial reference standards represented by the Japanese Pharmacopoeia Reference Standards.
Those reference standards are used in tests specified in the Japanese Pharmacopeia and other official compendia, and by providing
the basis for guaranteeing the reliability and objectivity of those tests, those reference standards help ensure and improve
the quality of pharmaceuticals and other products in Japan.